I’ve decided to take this blog more in the direction of communicating science – there are only so many sociological musings to come up with. This is the first of many planned posts explaining basic climate science so people have better context for what they read in the newspaper.
Every post is a work in progress, and will be continuously edited when necessary, so please leave comments with suggestions on how to improve the accuracy or clarity. Enjoy!
What determines the temperature of the Earth?
The temperature in your backyard, the warmth of the equator, the frigid polar regions, the average global temperature for the whole planet…..they might seem like very different things to measure, but they’re all caused by the same process. It all comes back to energy.
This energy comes from the Sun, but it’s not as simple as a single transfer. Remember, at any time of the day or night, the Sun is shining on some part of the Earth. That energy can’t just stay on our planet, otherwise it would keep building up and up and we would fry after a couple of weeks.
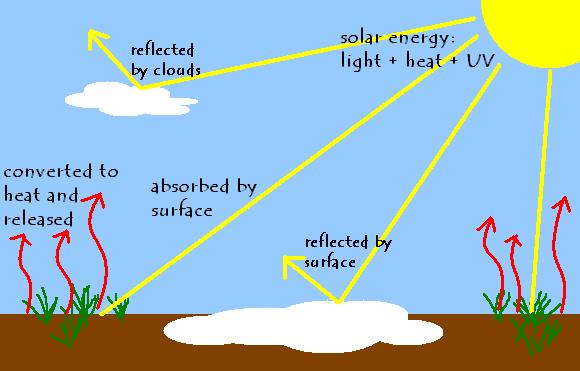
Therefore, incoming energy from the Sun has to be balanced by outgoing energy from the Earth for the planet’s temperature to stay relatively constant. So when the Sun’s rays hit the ground, as a mixture of light, infrared, and UV radiation, the Earth absorbs the energy. Then it converts it to all to infrared radiation, which we perceive as heat when it hits us, and releases it upward.
All objects perform this absorption and emission when they are hit with radiation. If they receive enough energy, they can release some of it in the form of light – think of how a stove element glows when it’s turned on. However, the energy hitting the Earth is nowhere near this level, so it all comes out as infrared.
It is this emission of infrared radiation that determines the temperature of the Earth. The second step, not the first, is the important one, the one that we actually feel and experience. So on a hot summer’s day, it isn’t actually energy coming down from the Sun that’s making the air warm. It’s energy coming up from the Earth.
The air doesn’t warm up instantly, either – there’s a bit of a lag. This allows warm air to be transported away from the Equator and towards the poles, in the global circulation system of wind currents. Without this lag time, many regions of our world would have far more extreme temperatures.
Additionally, not all the radiation the Sun sends down gets absorbed by the Earth. Some of it is bounced back by clouds, which is why sunny days tend to be warmer than cloudy days. Some of it reaches the surface of the planet, but is bounced back too, before it’s even absorbed. This reflection of energy is particularly common when the surface is light in colour. That’s why it seems so bright outside after a snowstorm – because the snow is bouncing the energy back up as light, instead of absorbing it and releasing it upward as heat. It also explains why dark concrete, which absorbs almost all the radiation that hits it, is so much warmer than a light-coloured deck.
The amount of energy that the Sun sends down to us is greater than the amount that the surface of the Earth actually absorbs. However, the amount absorbed has to be equal to the amount released, and the amount released is what we witness as the temperature outside.

Minor pedantry:
‘That’s why it seems so bright outside in the winter’ – you’re making an assumption that there’s always snow on the ground in the winter. Yes, you go on to make it clear that this is what you’re referring to, but the prose in its current form could confuse the reader.
Well, it confused this one, temporarily, anyway :)
Thanks for that correction. Wherever you live where there isn’t snow in winter, I am jealous! -Kate
Temperature is an interesting concept. We sometimes take the numbers literally.
Such as the thermostat setting on a central heating system. You can set it to 18 degrees C, but the reality is the amount of work required to reach that temperature depends on the temperature outside (your energy budget issue). So just adjusting the thermostat one degree can make an enormous difference in energy used even though the change in the setting is small.
Of course if the temperature outside is significantly lower (like your Canadian winter) then a small thermostat change might not save much energy.
We had some snow this year where I live, but it wasn’t for very long, but it did cause a lot of chaos. That’s the South of England for you!
On the energy budget issue of the planet, what is happening is the energy from the Sun comes to us at a higher frequency, then gets knocked down in frequency when it gets bounced back out towards space (or gets absorbed by the GHGs).
Thank goodness we aren’t bathed in gamma radiation!
There is apparently a star (can’t remember the name/number) which could shoot a huge burst of gamma radiation at us in the next thousand years or so, but apparently recently they worked out the beam would be pointed at a slightly different angle than was originally thought and will miss us by a few light years if/when it happens. Lets hope they have calculated the angle correctly the second time!
Intermittent drizzle is so much better than snow, right? :)
I have to say, Kate, that your new direction for the blog is arguably a bit redundant to existing efforts. Of course anything would be somewhat redundant, but one thing I’d like to see more of is interviews with working climate scientists that not only discusses their findings in a more rounded manner than one typically sees in media coverage but covers the basics of their methods. You could arbitrarily limit the potential subjects to Canadian scientists to keep things from getting too open-ended, plus it would probably make them more likely to want to respond to you.
I will boldly mention a few ideas:
1) Andy Weaver. What’s involved with ramping up his model for the AR5 and in particular incorporating the carbon cycle?
2) David Barber. The subject matter is obvious, but the content of the media coverage seemed even thinner than the usual. What’s happening now? What are the plans for future obs?
3) The MPB. I’m not sure who the lead scientist is, but from a recent look I had around the internet the beetles are established around Slave Lake and appear poised to make the jump from the hybrid pine zone into the jack pine-dominated boreal forest proper. I remember seeing several comments over the last few years that it wasn’t clear the MPB would do well on a jack pine-only diet, but from what I could see that’s always been just wishful thinking.
Finally, given the high quality of your posts and what I imagine must not be the best traffic here given the posting frequency and the relative newness of the bl;og, maybe you could find someone with a more established blog who’s willing to re-post at least some of your work.
That is a good idea and certainly something to think about. It would be lots of fun to interview scientists! Perhaps I could take my blog in two new directions at once. -Kate
Steve Bloom. One of the reasons that there is any debate at all about climate science is because of poor communication between scientists and the wider public.
I have encountered it myself (i have a background in engineering), I have come across academics that have made statements in texts and videos etc. that if you had sufficient knowledge would make sense, however without that knowledge can sound ridiculous.
I recently came across an engineering professor that made an innocent comment in a video that made it sound like his invention would be hugely expensive. I didn’t think about it first because I was more interested in the engineering/science, but when I thought about it, the costs expressed in the video did sound ridiculous. Anyway, I eventually emailed him to ask why his invention would be so costly, he then questioned how I arrived at the big numbers and he explained in more detail. Anyway what he was saying clicked and it was clear the opposite was true and his invention would be cheaper than similar technologies!
But his poor description made it sound expensive.
It wasn’t just me that thought his invention was expensive because of his poor communication, a few other people did as well.
The same goes for climate science, scientists are often not interested in explaining the basics starting from first principles (eg. quantum physics).
Yet it is these basics that explain the difference between O2 and CO2 in climate science and it is this sort of basic stuff that the ordinary person puzzles about, wondering why CO2 is so important whilst O2 isn’t. The denialsphere uses this lack of understanding to spread rumours and disinformation. Any hole in a persons understanding of anything can be exploited by the unscrupulous.
Although I agree with Steve Bloom, many people have done the basics before, there is nothing like putting it all down on paper to clarify your thoughts. You think you have a good understanding of something, start to put in paper and it turns to mush. Tiny holes become gaping chasms.
There is also the possibility you will come up with a way of explaining that gets through to some of those who do not understand. Or one of the commentators will correct something that others also have misunderstood.
Recapping, done before but still worthwhile.
Just a clarification – the new focus on science is in no way replacing any of the topics I currently write about. Rather, it’s an addition, as my way of exploring new directions, as well as actually contributing to science communication instead of just talking about how important it is. So if you like the blog the way it’s been going, no worries – you’ll just be getting even more. -Kate
Hi there,
Good explanation. I’m still struggling with certain aspects of climate science, including the energy budget. A question: We have an equilibrium between ingoing and outgoing flux leaving a temperature of X on the planet. Then due to some forcing or other the outgoing flux drops leaving the temperature of X+n at a new equilibrium.
But the thing I don’t get is considering this n extra heat will be passed through the atmospheric layers up to the stratosphere (now cooled from the GH effect), why doesn’t it get transferred out again. As space is now ‘cooler’ in relative terms shouldn’t this extra heat be radiated more?
James
Kate,
While infrared radiation is often called “heat radiation” this is confusing. Infrared radiation is radiation of a certain frequency (photons). Heat is the vibration of massy particles (kinetic energy).
Objects emit radiation in a band of frequencies corresponding to their temperature. Most everyday objects emit in the infrared. Electric heaters emit in the red; most wood fires tend to the orange, and the core of a bunsen burner burns blue; this change in frequency indicates increase in temperature. But the radiation itself is not heat.
I had this confusion myself in high school. I think it is endemic to high school teachers. Lose it, please.
The earth emits in the infrared because it is at a temperature where the radiation is in the infrared. The earth is negotiating with its surroundings to reach a radiative equilibrium, where incoming and outgoing radiation balance. This is what sets the temperature.
Only radiation travels through space. No “heat” arrives from the sun without being converted to radiation.
Another point you miss is that air does not heat or cool instantly; consider why a north wind is cooler than a south wind in your part of the world.
And…
mt
Thanks for the suggestions, mt. I made some changes, especially to the heat/infrared thing. What seems to be the easiest way to explain something can end up causing confusion later on. Sort of like the concept of electron orbit shells…-Kate
James, the outgoing flux at the top of the atmosphere does reach a balance with the incoming flux at the top of the atmosphere. Or indeed, at any other well-defined surface.
But the relationship between the temperature profile of the atmosphere and the conditions of the surface is one more thing missing from Kate’s description. If the atmosphere becomes less transparent to infrared, the ground “sees” the middle atmosphere more than before. Since this temperature is higher than that of space, the ground warms up a bit. But everything gets into balance.
It is, for many purposes, a good approximation to think of the system being in radiative equilibrium, comparable to the way most undergrad thermodynamics problems assume a near-equilibrium even though the situation changes over the course of the calculation.
Thanks, mt! -Kate